Historic moment: Optogenetic methods restore partial vision in a blind patient

An international research team has shown that optogenetic therapy has helped to partially regain visual function in a patient with retinitis pigmentosa. This is a milestone towards a gene therapy that could restore vision.
Clinical trial results show for the first time that optogenetic methods can partially restore vision in a blind human patient.
The achievement marks a milestone towards developing mutation-independent therapies for inherited photoreceptor diseases. It was reported in Nature Medicine by an international research team led by José-Alain Sahel and Botond Roska including members from the Institut de la Vision and Hôpital National des Quinze-Vingts, Paris, the University of Pittsburgh, the Institute of Molecular and Clinical Ophthalmology Basel (IOB), StreetLab, and GenSight Biologics.
“Enabling a patient to regain partial vision by optogenetics could not have happened without the engagement of the patient, the efforts of our multidisciplinary team at the Institut de la Vision and GenSight, and the longstanding collaboration with Botond Roska, which was at the origin and core of all this project,” says first and corresponding author José-Alain Sahel, Distinguished Professor and Chairman of Ophthalmology at the University of Pittsburgh, Professor at Sorbonne University and Hôpital National des Quinze-Vingts, and Founding Director of the Institut de la Vision.
The findings provide proof-of-concept that using optogenetic therapy to partially restore vision is possible
, says last and corresponding author Botond Roska, Founding Director at IOB and Professor at the University of Basel.

Roska stressed all optic misfunctions originate in the eye or in the retina, which has an extremely important task in this process. It is practically a biological computer that records a video, creating 30 representations from this recording and forwards it to the brain. The brain, of course, does not see what is out there therefore it predicts, relying on these video captures what is in the outside world, he explained how the retina works.
There are 100 tiny parts inside the retina that do the computation. You should imagine it as a hamburger, the lower section of which is a layer of ganglion cells that communicates with the brain.

The team of researchers were focusing on retinitis pigmentosa which is a neurodegenerative eye disease where loss of photoreceptors can lead to complete blindness. This is responsible for 20% of all blindness. In this case the light-sensing layer ceases to be sensitive to light; it's as if we removed the upper layer of the hamburger, the top of the bun. The goal was to restore this light-sensing layer, explained Roska. It seems to be a trivial idea, but it required an immense technological advancement, and we had to develop everything ourselves, he added.
Optogenetics involves genetically altering cells so that they produce light-sensitive proteins called channelrhodopsins. The technique has a nearly 20-year history in neuroscience but, so far, clinical benefit of optogenetics had not been demonstrated.
A collaboration between the teams of José-Alain Sahel and Botond Roska culminated in the findings reported today that reflect 13 years of multidisciplinary effort.
The goal of the research is to treat inherited photoreceptor diseases, which are widespread causes of human blindness. Photoreceptors are light-sensing cells in the retina that use proteins called opsins to deliver visual information to the brain via the optic nerve. Photoreceptors progressively degenerate, and then blindness sets in. To restore light-sensing capability, the team uses gene therapy methods to deliver channelrhodopsins into the retina's ganglion cells.

Retinitis pigmentosa (RP) is a progressive, inherited, monogenic or rarely digenicblinding disease caused by mutations in more than 71 different genes (https://sph.uth.edu/retnet/sum-dis.htm). It affects more than 2 million people worldwide. With the exception of a gene replacement therapy for one form of early-onset RP caused by mutation in the geneRPE65 , there is no approved therapy for RP.
The professor also said gene therapy methods aiming for the retina's ganglion cells are the simplest of those currently available, but there are only better than this, and there are advanced researches with the new methods.

The patient
It was a long road until clinical trials, They started on mice that regained their vision and a gene therapy had to be worked out which was tried on monkeys.
Clinical trials were launched in 2018-2019. The first volunteer was a 58-year-old French man who began going blind 40 years ago. When the experiment started, he could sense light but could not distinguish shapes.
The patient received an injection in his worse eye of a harmless virus called an adeno-associated virus, which carried the gene for an opsin from algae.
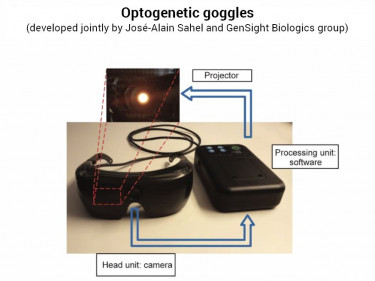
// For the current study, the team delivered the gene coding for a channelrhodopsin called ChrimsonR. This particular protein senses amber light, which is safer for retinal cells than the blue light used for other types of optogenetic research. The team also developed specialized goggles outfitted with a camera that captures and projects visual images onto the retina at 2/3 amber light wavelengths. //
The researchers waited 4.5 months for the ganglion cells in the patient’s eye to begin to produce the new protein. Then they began to train him to use special goggles that amplify incoming light from an image and focus it on the retina in the amber wavelength sensed by the opsin.
Within a few months, the man reported he could see the white stripes at a pedestrian crossing while walking outside wearing the goggles. He was “very excited,” although perhaps “not as much as we were,” study co-leader José-Alain Sahel, a physician-scientist at the University of Pittsburgh School of Medicine and the Vision Institute in Paris, said at the press conference.
Quantitative tests began when the man was able to see the white stripes at a pedestrian crossing, said Roska.
Thanks to this method, the entirely blind patient started to see objects. It is the first step in the process of restoring vision, and we hope that the events will pick up pace from now on
, he said.
Roska noted that they were able to go down this road only with a single patient because of the interruption by the pandemic. But there are other patients participating in the trial.
Roska showed video footage of the patient passing lab tests. First he had to find objects on a table, first a larger one then a smaller one, and he even had to count the objects. He could usually find and touch dark objects set on a white table, such as a notebook or box of staples, that he could not see without the goggles. He could immediately locate a transparent object and say what and where he sees it, and he could count up to three glass tumblers.


When the patient wore an electrode-studded cap that measures brain activity, the signals showed activity in the visual cortex, the part of the brain involved in seeing. The entire experiment involved two sessions, with a total of 183 randomized object/no-object trials for a total duration of about 140 min.

The team also trained a software decoder to evaluate the EEG readings. By simply measuring neuronal activity, the decoder could tell with 78% accuracy if the tumbler was present or not in a given trial. This last evaluation, Roska says, helped to confirm that brain activity is indeed related to a visual object, and "therefore that the retina is no longer blind."
Roska said he has never met the patient and he does not want to in order to remain emotionally detached in the treatment. He admitted that he saw data on the patient before he saw the video.
He did, however, deliver a message from the patient:
To those that are mourning over the loss of their vision I can say that based on the current result this treatment is filled with hope.
“Importantly, blind patients with different kinds of neurodegenerative photoreceptor diseaseand a functional optic nerve will potentially be eligible for the treatment. However, it will taketime until this therapy can be offered to patients” commented Sahel.
Roska said he sees two directions from here on out.
the first is that with the publication a new science, , visual rehabilitation, has been created.
He stressed that the brain needs a lot of time to learn the new language of the eye, because this methodology is not sending the same patterns as in a healthy person. We also need to learn how to teach people to see, he added.
The other direction is that there are various optogenetic strategies, and there are much better therapies than the one presented here. These are also works in progress.
Roska promised they would never give up. Illustrating the process of the research he said it's like if they were thrown into the ocean to seek out islands. We start in one direction and if there's nothing there, we swim back. We make mistakes all the time, but we know one thing for sure: we are never giving up. I'm sure we'll hit the wall hard a couple of times, but we'll keep going until we make one step forward, he allowed a glimpse into researchers' mindset.
Cover photo: Getty Images











